The physical universe includes two components: energy and matter. In most physical processes, there is a constant interaction between energy and matter. A basic law of the universe is that matter can neither be created nor destroyed. However, it can only be converted from one form to another.
Energy
The scientific explanation for energy is the ability to do work. People in the modern world have learnt to convert energy from one form to another. Energy can be classified into many different forms such as heat, light, motion, gravitational, chemical and electrical. These forms of energy can be converted from one form to another. For example, the kinetic energy stored in water can be converted to electrical energy, which in turn can be converted to heat and light energy. In our daily life, we use energy to walk, drive cars, cook food etc. The different forms of energy are grouped into two types: potential and kinetic energy.
The potential energy is the stored energy, and the kinetic energy is the working energy. The greater the kinetic energy associated with the body, the faster it moves.
Radiation
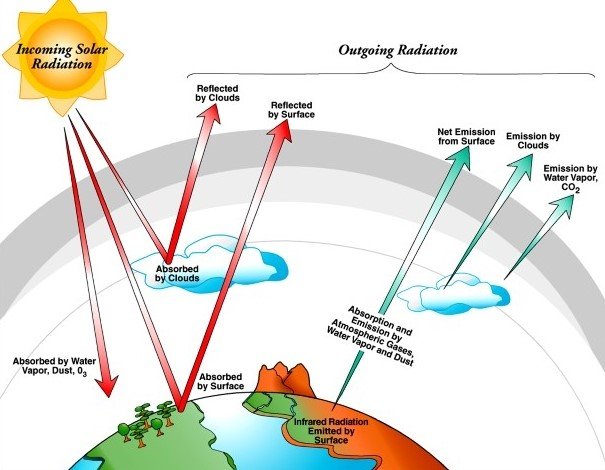
Radiation is a form of energy that moves from one object to another object through space. The speed of motion of the radiation is equal to the speed of light in a vacuum. Most forms of radiation can penetrate through a certain amount of matter. Examples of radioactive sources are the sun and other radioactive materials. The radiation can also be called electromagnetic waves. In nature, we can find a wide range of electromagnetic waves.
Radiation can be either ionising or non-ionising, depending on the effect of the radiation on matter. The non-ionising radiations include heat, visible light, radio waves and microwaves. These non-ionising radiation will not have enough energy to break the molecular bonds and remove the electrons from the atom. The radiation or electromagnetic waves with the highest energy, like ultraviolet rays, X-rays and gamma rays, can ionise the atom. The energy of X-rays and gamma rays is so high that when it interacts with atoms, they will remove electrons from the atoms, and it gets ionised.
Ionising radiation has a number of applications like cancer treatment, sterilising medical instruments and smoke detectors.
Alpha particles
Alpha particles are formed by the decay of heavy radioactive elements such as uranium, radium and polonium. The alpha particles are positively charged and made from two protons and two neutrons from the atom’s nucleus.
Beta particles
The beta particles are emitted from naturally occurring materials such as strontium-90; they are similar to electrons. These beta particles are used in the treatment of eye disease.
Uses of Radiation
1. Electromagnetic radiation is used in all forms of communication systems.
2. Radioactive atoms are used in carbon dating
3. Radiation is used in medical treatment for cancer treatment, diagnosis and research.
4. Radiation is used in determining the composition of materials. However, radiation also has an extremely harmful effect on living beings. They can cause burns, or in serious cases, cause cancer or other genetic disorders.